Hazardous breathing conditions exist in routine industrial operations, such as hospitals, abrasive blasting, paint spraying, industrial cleaning, and arc welding. In these and other operations that introduce contaminants into the workplace, supplied-air respirators are frequently used for worker protection. These applications utilize “low pressure” air (normally between 90 and 125 psig) and are designed to meet with OSHA and CSA breathing air quality requirements. This article outlines breathing air requirements and describes common applications, identifies commonly-found contaminants, and outlines a five-step contaminant removal process.
Breathing Air Requirements
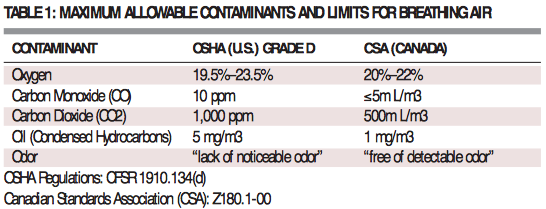
OSHA and CSA provide specific air quality requirements to provide worker protection. Individual industries, like the hospital industry, may have more specific requirements which provide specifications as to the type of machinery and alarms required to achieve breathing air quality – reliably and consistently. The breathing air requirements are set forth in the table below.
Hospital Applications
Hospitals use purified air for such processes as respiratory therapy, hyperbaric oxygen therapy, life support systems, and neonatal infant care. Patients may be using breathing air through various types of respirators, from half-face respirators to “iron lungs” in situations where the patient cannot take breaths on their own. Due to the critical, life-dependant nature of these installations, rigorous adherence to the 2005 NFPA 99 Specification (National Fire Protection Association) is practiced. Reliability, of course, is the primary objective in this situation. Specifications for equipment, alarm systems and back-up systems, which ensure breathing air quality and reliability, are detailed in the specification.
Abrasive Blasting Applications
Abrasive blasting operations most commonly use sand to clean iron and/or steel surfaces. This technique is also used to clean other materials such as brick, stone, concrete, sand castings, aluminum, brass, copper, wood, glass, and plastic. Silica dust (SiO2) is created when sand is used as the abrasive material. In terms of worker health, silica dust is of special concern because it may cause silicosis, a lung disease which develops slowly and often leads to death years after exposure has ceased. Dust is also a very serious health hazard in this industry. Dust is created by broken-down abrasives, pulverized surface coatings and encrusted substances, and abraded material from the object being blasted.
 |
| Abrasive Blasting |
Paint Spraying Applications
Paint spraying operations are subject to ambient air hazards presented by the common presence of organic vapors and mists from paints, lacquers, enamels and solvents. Please note that it is common, in this industry, to use only disposable, particle-type, face-masks. These face-masks, which afford only dust particulate protection, are commonly selected based upon comfort and not upon their ability to provide safe respiratory air.
Industrial Cleaning Applications
Tank cleaning and other industrial cleaning operations require solvents which can pose threats to worker safety. While the most common result of contact with organic solvents is dermatitis (inflammation of the skin), inhalation of solvent vapors will have more grave effects since all organic solvents may have some effect on the central nervous system. Trichlorethylene and perchloroethylene, two of the more toxic solvents, are used extensively in the vapor degreasing of metals and equipment. Known effects of exposure to these solvents include dizziness, headaches, loss of inhibitions, lack of coordination and central nervous system depression.
Arc Welding Applications
Arc welding processes create metal fumes and gases, which cause health concerns. Ozone and the oxides of nitrogen are the principal toxic gases produced by the arc welding process. Other contaminants may arise from the various components of the welding rods, oxides of the metals and the alloying elements being joined. Ozone, an intensely irritating gas, is produced by the action of the electric arc through air. Some paints may produce toxic fumes when heated with the welding torch. Other toxic fumes are generated when the welded metals contain, or are coated with, alloys of lead, zinc, cadmium or beryllium and are capable of producing metal fume fever. Symptoms usually occur a few hours after exposure and are similar to those of influenza: a metallic taste in the mouth, dryness of the nose and throat, weakness, fatigue, muscular and joint pain, fever, chills and nausea. These symptoms usually last less than 24 hours after which follows a temporary immunity. This is why welders are more susceptible to this condition on Mondays or on workdays following a holiday.

Common Compressed Air Contaminants
Supplied air respiratory protection systems originate with oil-lubricated or oil-less air compressors. These air compressors are placed in a multitude of different ambient air conditions from which they compress air. Ambient air may contain carbon monoxide, water vapor, oil, and dirt – to name a few of the contaminants. Identifying and removing these contaminants is the function of the system.
Carbon Monoxide
Carbon monoxide combines readily with the hemoglobin in red blood cells, rendering them incapable of carrying oxygen to tissues. Called anoxia, it causes dizziness, loss of motor control, unconsciousness and, in extreme cases, death. Since hemoglobin takes up carbon monoxide about 200 times as fast as oxygen, even low concentrations are very dangerous, and anoxia can develop even though the oxygen supply to the lungs is ample.
Carbon monoxide originates from ambient vehicle exhaust and other sources of hydrocarbon combustion. Air compressors, exposed to this type of ambient air, will send it downstream. Air compressors, supplying breathing air, must therefore have their ambient air intakes installed in a clean ambient air environment, with reduced ambient contaminants and normal concentrations of oxygen. Carbon monoxide can also be produced by overheated conditions in the air compressor itself. OSHA requires high temperature alarms on lubricated air compressors because overheated conditions can produce carbon monoxide by the partial oxidation of oil and oil vapors.
Water/Water Vapor
Ambient air contains moisture, which is drawn into the air compressor and is entrained in the airstream in a vapor state. As compressed air flows through the breathing air system it cools, causing water vapor to condense in face-pieces and helmets, obscuring vision. Even in small concentrations, moisture combines with oil and solid contaminants in the system to form sludge, which can clog or damage critical components, such as pressure regulator valves. It also causes rust in pipelines, that increases wear on breathing air components. Moisture can freeze in air lines, exposed to cold weather, and partially or completely block the flow of air. This is of particular concern to workers, who work outdoors, where their air lines are exposed to freezing temperatures.
Oil/Oil Mist
In systems using oil lubricated air compressors, oil is a major potential contaminant in the system. In reciprocating compressors, lubricating oil applied to cylinders is fragmented into fine particles by the shearing action of the piston and enters the airstream in mist form. In rotary screw compressors, oil is injected directly into the compressor chamber, and although most of the oil is removed by a separator, some ultimately enters the air stream.
In addition to the risk of producing carbon monoxide, in an overheated lubricated compressor, oil in mist form can present other problems. Although of limited toxicity, oil mist can cause breathing discomfort, nausea, and in extreme cases, lipoid pneumonia. It can also create unpleasant tastes and odors which interfere with a worker’s capacity and desire to complete a task. Oil can also act as a chemical corrosive and accelerate the deterioration of gaskets and seals. Oil, when combined with water, can also form a sludge which can clog pneumatic valves and controls.
Solids
Solid particulates have adverse effects on workers and can cause breathing difficulties. This can increase the chances of allergic reactions and, in extreme cases, cause sympton similar to those of silicosis. Solids generally enter the system through the air intake. In a typical metropolitan area, concentration of atmospheric solids may be as high as four million particles per cubic foot of air. When air is compressed to 100 psig, this concentration can be eight times greater. In some systems utilizing non-lubricated compressors, however, it is possible for the air compressor to introduce solid particulates. Teflon, carbon, and other materials are used in place of oil as a lubricant. Frictional wear on these compressor materials can cause particles to enter the airstream.
Five-Step Air Purification Process
Breathing air purifiers follow a five-step process to eliminate contaminants and to provide OSHA Grade D Breathing air quality.
- Liquid Oil is removed from the air stream with a coalescing filter. Compressed air flows into a blended-fiber filter element which removes oil droplets to 0.01 ppm.
- Water vapor and liquid is removed by a compressed air dryer. Depending upon the system specifications, a refrigerated dryer or a desiccant air dryer may be used. Dew points may be achieved, depending upon the dryer type, between 32 F and -40 F. Desiccant type dryers may use one or two beds of desiccant. Desiccant materials used are typically activated alumina, silica gel, or molecular sieves, which have a high capacity to capture and retain large quantities of moisture. Continuous-duty applications will normally use a dual tower desiccant dryer which can regenerate one desiccant bed in one tower, while the other tower dries the compressed air. An electric timer, or a sensor, will automatically cycle the air flow through the desiccant beds to provide a continous supply of dry air.
- A catalytic converter converts carbon monoxide (CO) to carbon dioxide (CO2). The bed of catalyst oxidizes the CO to CO2, drawing oxygen from the air. The reaction is: 2CO + O2 → 2CO2. The conversion rate decreases markedly when the air contains moisture or other contaminants. Since air from a compressor or an aftercooler is generally at or close to 100% relative humidity, the relative humidity must be lowered before the air passes through the catalyst.
- Oil mist (or hydrocarbons) are now removed from the compressed air stream with a filter which uses activated carbon to remove this taste- and odor-causing contaminant. Although not toxic, tastes and odors can interfere with worker comfort and performance.
- Solid particulates are filtered out by a final filter. Entrained particulates and desiccant dust are removed in this final stage of air purification.
For more information visit http://www.spxflow.com.