how the latest technologies can help a plant meet SQF guidelinesAny modern food manufacturing facility employs compressed air extensively in the plant. As common as it is, the potential hazards associated with this powerful utility are not obvious and apparent. Food hygiene legislation to protect the consumer places the duty of care on the food manufacturer. For this reason, many companies often devise their own internal air quality standards based upon what they think or have been told are “best practices.” This is no wonder, as the published collections of Good Manufacturing Practices (GMPs) that relate to compressed air are nebulous and difficult to wade through (Figure 1).
Understandably this has led to a significant difference in the quality of compressed air used throughout the food and beverage industry, with major differences even existing in plants owned by the same company. The goal of this article is to help make sense of it all. First, we outline the potential risks and hazards that compressed air systems can introduce to food products. We then benchmark published Good Manufacturing Practices as they relate to compressed air use in a food processing facility under a Global Food Safety Initiative (SQF, BRC, FSSC22000) environment. Finally, we provide several compressed air quality GMPs based on those published standards.
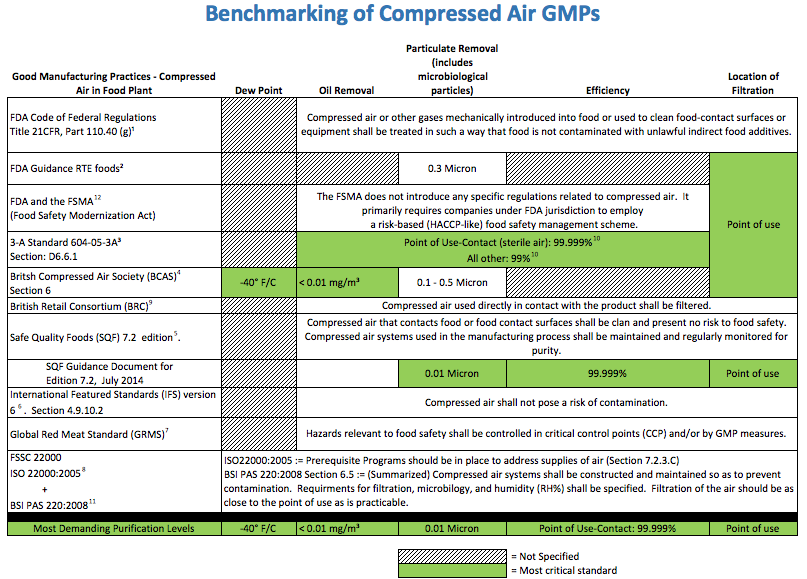
Figure 1: Published Good Manufacturing Practices related to compressed air can be difficult to understand and apply to a food processing facility. Click here to enlarge.
Knowing the Potential Risks of Untreated Compressed Air
Compressed air is not as clean as it appears to be. Untreated compressed air contains many potentially harmful or dangerous contaminants that must be removed or reduced to acceptable levels in order to protect the consumer and provide a safe and cost-effective production facility. Along with moisture and particulate matter, inlet air to a compressor generally carries 5 to 50 bacteria per cfm. A 75-hp compressor with a capacity of 300 scfm therefore takes in 100,000 to 1 million bacteria each hour. These bacteria get compressed along with the air and begin their journey through the compressed air system. Introducing this type of microbial contamination to food products is very risky and would be considered a lack of control by the facility. Understanding how to integrate the treatment of compressed air in a facility will help ward off that risk.
Where Does Compressed Air Contact Food?
Sometimes it is not apparent where the compressed air is contacting the food. Working surfaces like counters and conveyors are obvious and manageable contact points. Compressed air is invisible. It leaves no visible trace where it contacts the food, other food contact surfaces, or the packaging. Without adequate hurdles and physical barriers in place, the microbial, particulate, and (in some cases) compressor oil contamination is left behind after the air dissipates.
Some example applications that present direct and indirect contact points are:
- Bagging
- Sparging/Mixing
- Drying
- Air Knives (Blow-Off)
- Pneumatic Exhaust (i.e., cylinder exhaust)
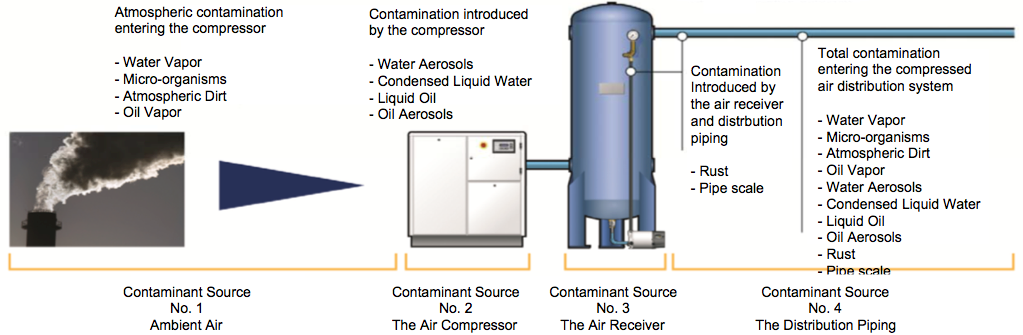
Without proper treatment, contaminants can travel from ambient air all the way through the compressed air system.
Safe Quality Food Standard: 5 Compressed Air Criteria - Webinar RecordingDownload the slides and watch the recording of the FREE webcast to learn:
|
Managing the Risks of Compressed Air in Food Processing
Compressor room drying and filtration are good, but they are not enough for a food processing plant. System filtration can do a good job reducing the amount of contaminants introduced into the downstream distribution system. However, that alone does not meet the requirements of the published GMPs that address compressed air — nor is it fully effective. In this scenario, the risk of food adulteration is still quite high. The warm, oxygen-rich environment inside the downstream air reservoirs, piping, fittings, and controls are ideal harborage sites for microbial biofilm growth — especially when fed with food-grade compressor oils that inevitably migrate downstream. For this reason, a number of the published GMPs call for point-of-use filtration that should be in place for all points where compressed air either directly or indirectly contacts food.
The first line of defense to ward off potential microbial contamination of the food product from compressed air is to use point-of-use sterile air filtration. With a properly designed compressed air system employing the benchmarked GMPs (outlined later in this article), along with well-designed Sanitation Standard Operating Procedure (SSOP) maintenance and monitoring programs, the risk associated with compressed air at points of contact can be mitigated significantly. A system design employing sterile air filtration at point-of-use puts a physical barrier in the air stream guarding against microbial contamination of the food. Combining this system design with an HACCP Prerequisite Program (PRP) formalizing these GMPs and SSOPs makes a cost-effective, efficient, and defensible risk management plan.
Compressed Air for Ready-to-Eat (RTE) Foods
RTE foods are at high risk of contamination from compressed air. Any microbial contamination introduced in the later stages of RTE food processing can stay with the food all the way to the consumer, as few hurdles or barriers are generally in place to eliminate the hazards. Point-of-use sterile air filtration is critical to ensuring RTE food safety at any point where compressed air can contact the food or food contact surfaces.
 |
 |
Parker Balston 3-Stage point-of-use filtration modules, in standard and 304 stainless steel housings, will remove contaminants at a very high efficiency - up to 99.99% for 0.01 micron particles and droplets. The final stage of filtration removes all viable organisms with an efficiency rating of 99.9999+% at 0.01 micron (5-log reduction). |
|
Preventing the Growth of Microbes – Benefits of Dry Air
The warm, dark, moist environment inside a compressed air system is the perfect condition for microbes to flourish and grow. Drying the air to a low dew point is an effective way to inhibit this microbial growth. Inhibit — not kill. Microbes need food, water, and the right temperature to grow. Take one or two of those nutrients away, and the growth stops — temporarily. Some of the microbial pathogens that are hazards to food safety form spores and/or protect themselves by moving into a dormant stage when nutrients in the surrounding environment are depleted. These dormant spores resume propagation as soon as the missing nutrients (moisture) become available again through contact with the food.
“Bacterial spores survive very dry conditions without any problem. Vegetative bacterial cells can survive dried states for a period of time. In fact, lyophilization (freeze drying) is a common way to preserve bacteria. Once conditions are favorable for growth (moisture, nutrients etc.), the bacteria can grow again. The foodborne pathogen Salmonella is notorious for surviving under water limited conditions.”14
The best practice for food safety is to first dry the air, and — more importantly — use point-of-use filtration to capture the microbes and spores, so they never come in contact the food.
Compressed Air Purification & Piping Monthly e-NewsletterWith a focus on Demand-Side Optimization, compressed air dryers, filters, condensate management, tanks, piping and pneumatic technologies are profiled. How to ensure system reliability, while reducing pressure drop and demand, is explored through System Assessment case studies. |
Monitoring Compressed Air for Purity
Whether it is specified or implied by the food safety scheme being employed in a plant, regularly testing the purity of compressed air coming in contact with food is a best practice. A single test at one point in time is not enough. Compressed air systems are dynamic and the compressor intake is subject to microbial, particulate, and moisture variations throughout the year — as well as buildup of contamination in the system.
In addition to testing at the compressed air’s point-of-food contact for pathogenic/allergenic contamination, it is also advisable to test the compressed air for yeast and mold spores where the product is packaged. Many companies find that yeast and mold spores from compressed air introduced in their final packaging operations can be one of the culprits impacting shelf life. Potato dextrose agar is best for testing for the presence of yeast and mold spores in the compressed air.
 |
 |
The Parker Balston Compressed Air Microbial Test Unit (CAMTU) is a lightweight, easy to use device capable of sampling compressed air systems for microbes. The unit requires no electricity and has a quick sampling time of 20 seconds. The CAMTU is an ideal device to incorporate into your Good Manufacturing Practices program for monitoring all identified HACCP risk points. Illustration of the CAMTU, Regulator, and Shutoff Valve to Sample Point (left to right). |
|

Unlike the conventional agar plate, this unique CAMTU agar plate offers greater dispersion of the compressed air over the agar as a result of an improved air flow path through the center hole in the plate. This provides optimum detection performance and enhanced capture of microbes.
Understanding the GFSI/SQF Environment
Due to the risks mentioned previously, the number of food manufacturing companies adopting GFSI-endorsed food quality schemes is steadily growing. One of the most popular schemes in the U.S. is the SQF Code. Beginning with the 7th edition of the SQF code, released in July of 2012, awareness of potential contamination from compressed air was highlighted. The 7.2 Edition of the SQF code, published March 2014, includes verbiage relating to compressed air in Module 11: Good Manufacturing Practices for Processing of Food Products. SQF has also published a Guidance Document to accompany the Edition 7.2 Code.
The key points applicable to robust GMPs are:
SQF Module 11.5.7.15: “Compressed air that contacts food or food contact surfaces shall be clean and present no risk to food safety.”
- “Wherever the compressed air comes in contact with the food, either directly or indirectly, high efficiency filters are to be in place at point-of-use where the air enters the final section of tubing (not in the compressor room).”
- "The recommended final stage of filtration in these food contact areas should have a rating of 0.01 micron with an efficiency of 99.999% (or as determined by appropriate risk analysis). "
- “It is generally advisable to locate the filtration as close as practically possible (near the “point of use,” or the point where air contacts the food), so as to not have long lengths of piping/tubing between the microbial removal filter and the air/food contact point. “
SQF Module 11.5.7.25: “Compressed air systems used in the manufacturing process shall be maintained and regularly monitored for purity.”
- “…[compressed air] testing must be conducted at a minimum of once a year.”
- “Testing can be done in-house or by a contracted party.”
- “Microbiological testing can include testing for aerobic plate count and/or indicator organisms as appropriate to the operation.”
- “Aseptic sample collection needs to be used.”
Parker GMPs for Point-of-Use Compressed AirTo help companies adhere to GFSI standards, Parker has developed recommended Good Manufacturing Practices (GMP) for compressed air systems in food processing plants based on benchmarking published practices. The following three simplified GMPs are recommended for food processing facilities using compressed air:
|
System Design GMP: Drying
To retard or stop the growth of microorganisms in the system, the pressure dew point should be reduced as close to -40°F/C as possible. A compressed air dryer should be installed to remove this moisture. Ideally, the dew point should be reduced to -40°F/C (ISO8573-1:2010 Class 2 for humidity and liquid water). Refrigerated dryers will provide dew points in the ~+38°F range (ISO8573-1:2010 Class 4 for humidity and liquid water). If a refrigerated dryer is used, it is important that the three stages of filtration mentioned below be in place.
System Design GMP: Point-of-Use Filtration
When designing a compressed air system, use point-of-use filtration wherever compressed air comes in contact with the food — either directly or indirectly. Point-of-use filtration is the best line of defense against microbial contamination of food in a compressed air system. Even the best compressor room system filtration does not eliminate harborage sites and biofilm buildup in the downstream compressed air piping system. The following three stages of filtration will significantly reduce the risk of microbial contamination of the food.
- Stage 1: Remove bulk liquid and particulate matter down to 0.01 micron at ≥ 93% coalescing efficiency.10 Automatic drain in filter. (ISO 8573-1:2010 Class 2.4.2)
- Stage 2A: Remove oil and water aerosols and smaller particulate matter down to 0.01 micron at ≥ 99.99% coalescing efficiency.10 Automatic drain in filter. (ISO 8573-1:2010 Class 2.2.2)
- Stage 2B (Optional): If there is a concern for hydrocarbon vapor carryover from the air compressor, then the installation of activated carbon filter may be necessary. (ISO 8573-1:2010 Class 2.2.1)
- Stage 3: Remove microbial contamination down to 0.01 micron at ≥ 99.999% particulate removal efficiency10 (5-log reduction) with a sterile air filter. (ISO 8573-1:2010 class 1.2.2. Class 1 – or better – particulate is key.)
Sanitation Standard Operating Procedures: Maintenance of Filters
- Stage 1: Change filter element every 6 to 12 months.
- Stage 2A: Change filter element every 6 to 12 months.
- Stage 2B: Change filter element every 3 to 6 months.
- Stage 3: Change filter element every 3 to 6 months — or sooner — as necessary based on point-of-use air quality test for microbial content.
- Note: Sterile air filters are designed to capture microbial matter larger than the nominal element rating. Microbial matter will not create a differential in pressure across the element. Therefore, measuring differential pressure across the element will not give an accurate reading of contamination. Air testing and/or regularly scheduled element changes are the best practices.
Sanitation Standard Operating Procedures: Monitor Purity of Compressed Air
Compressed air at 100 psig contains 8 times the amount of bacteria and contaminants as atmospheric air. In addition, Mesophilic Aerobic bacteria and fungi love the warm dark environment inside a compressed air system. As a baseline, test compressed air at each food contact point at least annually. Determine the test interval empirically based upon presence of microbial contamination.
Following Compressed Air GMPs
Identifying the risks and potential hazards compressed air introduces in a food processing plant is the easy part. Determining — and following — Good Manufacturing Practices for effectively treating the compressed air is not so straightforward. In the end, the best and final defense against all types of compressed air contamination is point-of-use filtration.
For more information, contact Lee Scott, email: Lscott@parker.com, tel: (978) 478-2750, or visit www.balstonfilters.com. To read more about Food-Grade Compressed Air Standards, please visit www.airbestpractices.com/standards/food-grade-air.
Notes and References
³ 3A Standard 604‐05 may be purchased at: www.techstreet.com/3Agate.html
⁴ http://www.bcas.org.uk/compressed-air-codes-of-practice.php
⁵ http://www.sqfi.com/standards/
⁶ http://www.ifs-certification.com/index.php/en/retailers-en/ifs-standards/ifs-food
⁷ http://www.grms.org/GRMS_standard.aspx
⁸ http://www.iso.org/iso/home/store/catalogue_tc/catalogue_detail.htm?csnumber=35466
⁹ http://brcglobalstandards.com/Manufacturers/Food.aspx#.VnldTPFCZP4
¹⁰ For coalescing: as measured by ISO 12500-1:2007, Filters for Compressed Air-Test Methods-Oil Aerosols. For particulates: as measured by the Fine Test Method, ISO 12500-3:2009, Filters for Compressed Air-Test Methods-Particulates.
¹¹ http://shop.bsigroup.com/en/ProductDetail/?pid=000000000030181317
¹² http://www.fda.gov/food/foodsafety/fsma/default.htm
¹³ Food & Beverage Brochure – www.camfilfarr.com
¹⁴ McLandsborough, Dr. Lynne A., PhD, Associate Professor and Director of Undergraduate Food Science Program, Department of Food Science, University of Massachusetts, Amherst