The useful and various properties of nitrogen (N2) in industrial applications rank it as one of the most specified gases in industry. For the manufacturer, nitrogen options exist in the choice of delivery system, compliance with clean air standards, safety and purity1. In researching these choices, manufacturers can accurately select the optimum nitrogen supply required, often at a considerable savings. Selecting purity levels of 99.99% or higher in many industries and applications ads a variety of costs, both financial and efficiency, which may be needlessly incurred.

Parker dh Multibank Nitrogen Generators at a Coffee Packager
Commercially Supplied Nitrogen: The Process and the Costs
Liquid air separation plants provide nitrogen generated by using cooled air to separate out the oxygen and nitrogen as they become liquid. Cryogenic distillation accounts for approximately >95% of the total nitrogen production. Generating nitrogen using this method is energy-intensive because the process entails condensing ambient air into liquid air by cooling and compressing it in a refrigeration cycle that utilizes the Joule-Thompson effect.
After N2 is separated from the air, additional energy is needed to purify it to requirements and fill the appropriate transport container. Since this process is performed continuously on a large scale, its power usage generates hundreds or thousands of tons of greenhouse gases every day.
Nitrogen produced through this process can attain a purity of 99.99% or higher. This process is completed at a higher cost for a purity level offering no added benefit for most applications. Purchased nitrogen from a commercial gas company is the most expensive option. Costs are slightly lower for liquid nitrogen in a Dewar or bulk tank. In cylinders, the cost rises.
The cost of transport of nitrogen via delivery tankers from a fractional distillery facility to and from an end users plant is factored into the price. Delivery of nitrogen uses a lot of energy and significantly contributes to the amount of CO2 generated in the process of delivering nitrogen to end users. Obviously, the amount of energy required to transport the nitrogen depends on the distance between the facility and the end user’s plant, but the environmental impact of trucking nitrogen is significant. For example, a tractor trailer traveling around 100,000 miles per year generates about 360,000 pounds of carbon dioxide during that time period. The highly pressurized and heavy cylinders require proper handling and can lead to back injuries or catastrophic damage and injuries if the valve were to shear off.

Parker NITROSource nitrogen generators
On-site Nitrogen Generators: The Process and Benefits
On-site nitrogen generators are safer and easier to handle than high-pressure cylinders and offer speed of delivery advantages over liquid nitrogen evaporation from dewars and tankers. On-demand gaseous nitrogen generation uses one of two alternative methods. They include Pressure Swing Absorption (PSA) and membrane system technologies. The choice of generator largely depends on the purity of nitrogen needed. In both cases, the level of O2 can be controlled to just the required purity level.
Applications that need nitrogen of 95 to 98 percent purity (5% to 2% oxygen), such as fire and explosion prevention, can use membrane generators. Applications such as the blanketing of oxygen sensitive compounds, specialty chemicals and pharmaceutical processing need a high purity stream and require the use of PSA generators.
PSA systems take compressed air and filter the air using high quality coalescing filters to remove 99.99% of 0.01 micron particles. The system then separates nitrogen from oxygen based on the preferential adsorption and desorption of oxygen, water vapor and other contaminants on a Carbon Molecular Sieve (CMS) bed. PSA nitrogen generators have two bed columns: one with fresh CMS for the current adsorption process and another that swings to low pressure to desorb oxygen from saturated CMS. One column provides nitrogen while the other column is being regenerated. These column beds are cycled back and forth to generate and regenerate the beds. The resulting high purity nitrogen then feeds a buffer tank to allow for a continuous high-pressure output. The high purity nitrogen stream passes through a final filter to ensure the delivery of pure, sterile nitrogen.
The hollow fiber membrane system uses compressed air that is directed through the bore of a membrane tube. The smaller and more soluble oxygen and water molecules pass through the wall of the polymeric membrane tube thus enriching the nitrogen in the air stream. Membranes produce nitrogen in a continuous process assuring constant downstream purity. Like the PSA system, the nitrogen-enriched gas stream passes through a final filter to ensure the delivery of pure, sterile nitrogen.
Purity Specified in Industrial Applications
Although on site nitrogen generation can produce N2 purities of up to 99.999%, users can realize significant financial and energy savings if they match the purity of the nitrogen to the purity required by the application. Indeed, for many applications, a purity of 99.9% or above is not required.
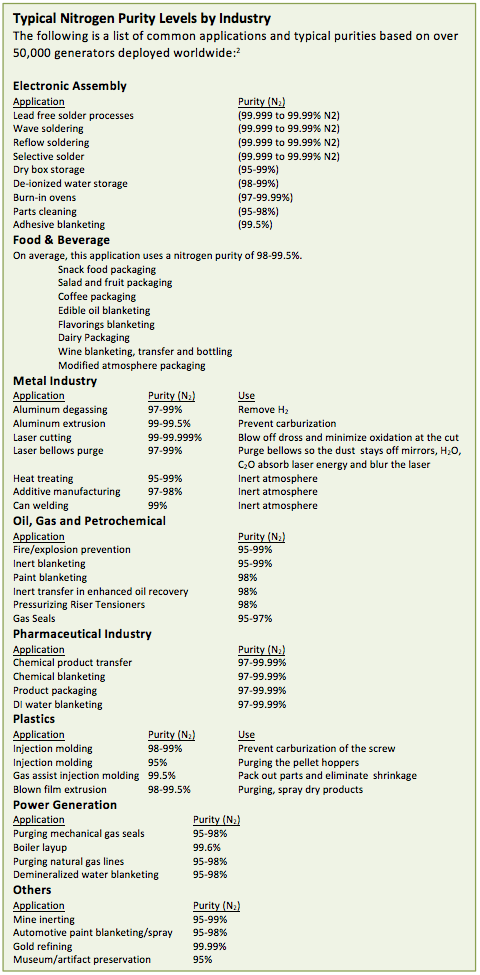
Nitrogen serves to extend shelf life and preserve flavor, color and aroma in food and beverage products ranging from snack food packaging, coffee packaging and wine bottling. A purity level between 98-99.5% is specified for most products. Packaging of many products is filled with nitrogen, an odorless and tasteless addition. Products can undergo longer shipping distances without a loss of flavor when the oxygen is replaced with nitrogen in the packaging. Nitrogen is occasionally mixed with CO2 or small amounts of O2 to create a modified atmosphere packaging to inhibit specific bacterial growth in meats, fish or poultry. Other products, such as edible oils are blanketed with nitrogen to prevent oxygen from inducing rancidity. In wine production, nitrogen is used both in the blanketing of the fermenting stock and in bottling. Bottles are purged with nitrogen, filled with wine and topped with more nitrogen before corked.
The metals industry values nitrogen for a wide variety of purposes. In aluminum degassing, the molten metal is sparged with nitrogen, pushing out hydrogen which can cause gaseous occlusions. In aluminum extrusion, the inert nitrogen prevents the formation of oxides. Laser cutting employs nitrogen to blow off dross and minimize oxidation at the cut, while a laser bellow is purged so the dust is removed from the mirrors and H2O and CO2 is cleared, preventing the absorption of laser energy and blurring the cut. Heat treating metal needs an inert atmosphere such as nitrogen. Purities range from 97% for aluminum degassing to 99.5% or higher for heat treating and aluminum extrusion. For laser cutting, purities of nitrogen can be as low as 99.95% or lower depending on the material and thickness being cut.
Oil and gas and the petrochemical industries face safety concerns, well served by the inert properties of nitrogen. Levels of purity in these industries are usually between 95-99%. Fire prevention and explosion blanketing successfully removes the oxygen with the introduction of nitrogen. Chemical tanks are often blanketed with nitrogen to prevent fire or explosion. In upstream and midstream areas of the oil and gas industry there are multiple uses, from the cleaning and inspection of pipes to the pressurization of riser tensioners, used to maintain stability in a floating or tethered rig. Within a pipeline, gas seal turbines are often sealed using a cushion of nitrogen. The nitrogen prevents natural gas leakage and suppresses the risk of fire should any slight leak occur.
The pharmaceutical industry relies on the inert properties of nitrogen to assure safety and sterility in the chemicals and packaging, with an average purity level of 97-99.99%. In product transfer, nitrogen is used to purge receptacles to eliminate contamination. Chemical blanketing, with nitrogen, assists with the stability of the final pharmaceutical product. Sealed packaging is infused with nitrogen to maintain freshness of the drugs. Deionized water, used throughout pharmaceutical processes, is blanketed to assure a constant pH by preventing exposure to CO2.
The development of plastics requires both the low dewpoint drying and inert properties of nitrogen for molding and extrusion processes. Nitrogen purities range from 95-99.5% in this industry. In injection molding, purging of the pellet hoppers and the prevention of carburization on the screw is achieved with nitrogen introduction. In gas-assist injection molding, pressurized nitrogen assists in packing out the part and eliminates shrinkage. Blown film extrusion uses nitrogen for both purging and spray drying products.
The Power generation industry is highly safety and maintenance conscious. Nitrogen assists in removing oxygen and preventing corrosion. During boiler cycling, the proper layup of the Heat Recovery Steam Generator (HRSG) includes nitrogen to avoid corrosion and pitting of the boiler, following ASME guidelines. A boiler that has been powered down is both blanketed and sparged with nitrogen to prevent corrosion. Nitrogen is often used to blanket and sparge demineralized water tanks to prevent CO2 contamination of demineralized water. Purging natural gas lines with nitrogen allows repairs or valve installations without fire concerns. Across the field of power generation, nitrogen purity is usually between 95-98%, with up to 99.6% in boiler layup.
In the Electronics Assembly industry, nitrogen is used to keep solder clean and soot free especially in the selective solder and wave solder processes. Purities in this industry can be as high as 99.999%
Other applications for nitrogen use are in diverse fields where its inert properties and purging of oxygen is needed. Coal or other mines can be partitioned off and filled with nitrogen to purge oxygen and prevent explosions. Automotive paint blanketing and paint spraying with nitrogen speeds drying and improves product finish. Museum artifacts and antiquities are often stored in a nitrogen atmosphere to preserve and protect their surfaces.
Conclusion
In the majority of industrial applications, a nitrogen purity of 99.9% or above is not needed to achieve the benefits nitrogen provides. Specifying a purity of that level may unnecessarily add to the cost of production, and the amount of energy needed to create and transport the nitrogen which impacts CO2 emissions. In the purchase of bulk nitrogen, manufacturers must consider safety in handling cylinders, the potential of delivery delays, additional truck traffic to and from the manufacturing site, as well as the environmental impact.
On-site PSA and membrane systems are energy efficient and cost effective, requiring only enough energy to power the air compressor that supplies air to the system. Gas industry sources indicate that an air separation plant uses 1976 kJ of electricity per kilogram of nitrogen at 99.9% purities. At a purity of 98 percent, the energy required for in-house nitrogen consumes 62 percent less energy. Even for those applications requiring 99.9% purity, generating nitrogen in-house on-demand with a PSA system will use 28 percent less electrically compared to third-party supplied bulk nitrogen.
1Nitrogen concentration or purity is defined in a percentage. The percentage represents 100% minus the oxygen content. The specified nitrogen is the inert gas content and will include Argon. For example, 98% nitrogen represents 2 % oxygen and the balance inert gases, i.e. nitrogen and argon.
2Based on observed industry averages and customer information feedback from Parker Hannifin Corporation Filtration and Separation Division installations worldwide in the listed industrial applications. Each customer should determine the nitrogen purity which best suits their applications.
For more information contact David Connaughton, Product Sales Manager, Parker Hannifin, Gas Separation and Filtration Division, email: dconnaughton@parker.com, tel: 978-478-2760
To read more Nitrogen Generation Technology articles, please visit www.airbestpractices.com/technology/air-treatment.