In modern and industrial work settings, people spend more than 90% of their time in enclosed spaces, such as warehouses, office buildings and factories. In most indoor environments, the air contains a variety of chemical and microbial particles, commonly defined as indoor pollutants, which can severely affect human health and product quality (1). Industries like food and beverage, medical devices and pharmaceutical manufacturers rely on their scheduled compliance testing to confirm the presence or absence of issues in workflow pipelines that are detrimental to the daily output and safety of the product. If contaminants are in the ambient air, whether indoors or outdoors, then you can be assured that they are also on your clothes, body, and equipment. A problem arises when certain pollutants like bacteria, yeast and molds cross contaminate regulatory and compliance tests like microbial testing of compressed air systems. Aseptic technique is a practice designed to thwart these effects.
The International Organization for Standardization (ISO) and the United States Pharmacopeia (USP) include in each microbial procedural section the use of aseptic technique while testing. The technique is used to avoid cross contaminating the tested compressed air sample with that of the pollutants on yourself and the surrounding environment. If not conducted properly, false positives will occur and can often result in system failures, accumulative costs, and maintenance or retesting fees. This article will focus on understanding the development, implementation, and importance of aseptic technique for compressed air microbial testing.
Evaluate the Sample Location
Understanding aseptic technique requires an understanding of how microorganisms interact with dynamic environments. These environments include the ambient air near the compressor and point-of-use, the sampling equipment, and finally the sampling technician themselves. Depending on where the compressor is located, it will pull in ambient air of variable quality. The system then compresses that air, sends it into a storage tank and then distributes it through a filter and dryer system (not required, but highly recommended) before it is used in the field. Once the air is distributed through a pipe or tube line to its point-of-use, the system can be tested for microbial contamination. If the environment around the point-of-use sampling area is filled with dust or other types of particulate matter, there is a large probability that those particles in the ambient air will cross contaminate the microbial test at the time of sample taking. For example, facilities that sort, clean, or package certain goods that result in a high degree of flocculate material like flour, sand, dust etc., can find that taking a simple microbiological test is next to impossible due to the surrounding environment. The particulate matter acts as a vehicle to the bacteria, yeast or mold and carries it to a new location. This can include the agarose plate used for sampling. Microbes are very small and can easily travel from one environment to another. Something as simple as a sneeze can send millions of bacteria across the room. Doing a risk assessment of areas with this high level of particulate matter in the ambient air is a great way to evaluate whether the point-of-use is a required point of testing. If the degree of cross contamination is very likely to yield inaccurate results due to the inability to sample with a certain degree of cleanliness, preloading the sampler in a more “clean” area might be necessary. This can help to achieve more reflective results of the point-of-use. Due to strict regulations and compliance procedures, pharmaceutical and medical device operations will likely not run into these types of issues.
Evaluate Yourself
The primary goal of aseptic technique is to eliminate the potential of cross contaminating microbiological samples with outside microbes. The biggest problem is that the human body is full of beneficial and potentially pathogenic microbes. These reside inside and outside the individual. A few simple pieces of personal protective equipment and some disinfectant can help prevent cross-contamination.
Gloves
The first and generally most effective pieces of PPE are clean, fitted examination gloves (mechanics or cloth gloves are not the same). Loose latex or nitrile from the fingers of a poor fitting glove can touch the surface of the agarose plate and contaminate it. Examination gloves produce an inorganic barrier between yourself and the sampling media (usually non-selective agarose). The Centers for Disease Control and Prevention states that keeping hands clean is one of the most important steps we can take to avoid spreading germs to others and multiple surfaces (2). Microbes can get onto hands after people use the toilet, handle raw meats, touch mucosal membranes, practice poor hygiene, and handle other contaminated objects (doors, knobs, tables). Protecting your compressed air microbial sample from yourself is the best way to ensure accurate results on your report. Once your gloves are on and sprayed with a disinfectant (ethanol, bleach or isopropanol alcohol), refrain from touching any surfaces other than the work surface. People generally touch their eyes, nose, and mouth without even realizing it. If there is a potential of cross contamination, change gloves or clean the surface of the gloves prior to moving forward.
Lab Coat/Apron
It is also important to consider your clothes and arms as a potential source of contamination. A clean, fitted, washable or disposable lab coat is another necessary form of PPE to eliminate any microbes that land or live on your clothes and skin from shedding onto the agarose plate during testing and movements during sampling setup. These lab coats need to be cleaned; a dirty lab coat/apron is as useful as no lab coat at all. Additionally, should there be a spill (alcohol or disinfectant) or the agarose plate is dropped on your body, the lab coat will keep you clean.
Facemask
People generally don’t realize how many microbes occupy the space in their mouths and nose. There are so many microbes in the human mouth that there is a study named after it: the human oral microbiome (3). A single mouth can be home to more than 6 billion bacteria, an impressive number when compared to the 7.3 billion people populating the Earth. Every time we speak, cough, sneeze, exhale a breath, saliva and microbes leave our mouth and nose. Most of these droplets degrade enough that the microbes can’t survive in them long, but studies have shown that after a sneeze, microbes can survive in the bioaerosol (the liquid of the sneeze or cough) for up to 45 minutes (4). Because of this, it’s important to cover your nose and mouth when opening microbial plates like agarose.
Disinfectant
Disinfectants are solutions that act to eliminate or reduce harmful microorganisms from objects or surfaces. This is different from a sterilization process, which kills all microorganisms. Alcohol has been used as a disinfectant for centuries. The most common of the alcohol-based sanitizers are ethanol and isopropanol alcohol. Due to the structure of some microorganisms, like bacteria, exposure to a solution of 70% ethanol or isopropanol alcohol will result in the outer cell membrane of the organisms to degrade, fall apart, and ultimately die. This does not happen as efficiently with alcohol concentrations above or below 70%. Solutions of 10% bleach (sodium hypochlorite) act like alcohol by degrading proteins of the microorganisms which results in cellular death. The simple act of spraying the surface of gloves, work table, and inside of the impaction sampler is enough to properly clean between points-of-use for accurate results.
These essential PPE items are necessary to avoid cross contamination when testing compressed air microbial samples using aseptic technique. If the technician has a beard or long hair, hairnets are always recommended. Eye protection is important for protecting eyes while using disinfectant sprays and preparing the compressed air for sampling.
Training Aseptic Technique
Click here to enlarge.
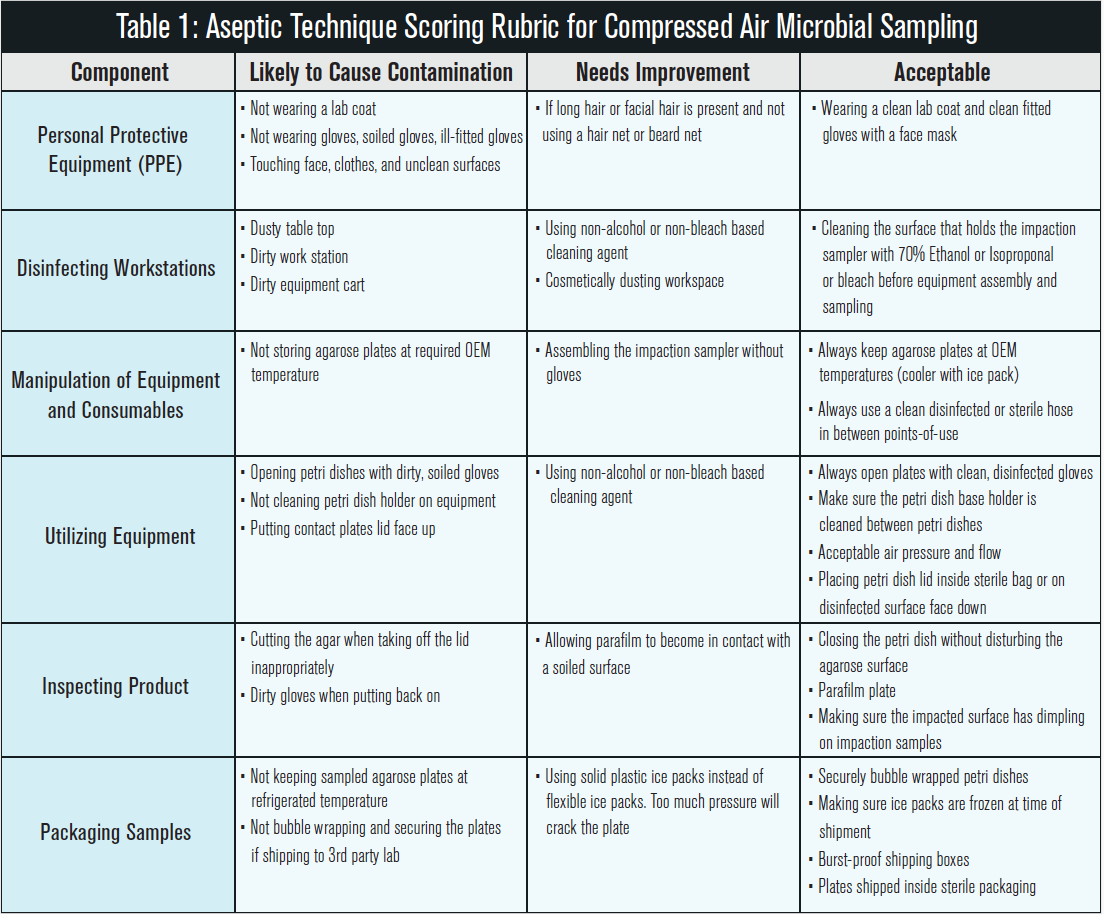
Adequate aseptic technique training includes many components. These include preparing the environment for testing, personal preparation, and product manipulation (5). Table 1 is an adaptation of Brown’s chart that outlines a training rubric for compressed air (impaction sampler) microbial testing in adherence to ISO 8573-7. The purpose of the training rubric is to allow for performance scoring and identification of particular areas to focus retraining on in the future. The table contains the component being evaluated, ratings based upon criteria descriptions, and their likelihood to cause contamination, need improvement or be acceptable in practice.
The components that need to be evaluated include personal protective equipment, disinfectant and cleaning protocol, manipulation of sampling equipment, utilization of equipment, inspecting final product, and packaging the final sample for shipment or processing. These all encompass the aseptic technique protocol that will limit or eliminate any cross-contaminating variables from the sampling equipment to the plate analysis. The rubric should be read in full and adapted to fit the sampling equipment being used. Much of the protocol is broad enough to be implemented universally and by all skill levels. It is recommended that a baseline of the technician’s skill be performed after an aseptic technique discussion is given and understood. This baseline will give the trainer an idea of what skills need improvement or concepts need clarification. As most cleaning and laborious protocols have degradation over time, it is recommended that a biannual assessment be conducted to evaluate technique over time.
Microorganisms in compressed air systems can indicate a potential presence of pathogens associated with wastewater near intake pumps, poor preventative maintenance of piping distribution, and loss of protocol integrity. Thorough, documented training is critical when it comes to making sure personnel are adequately prepared to sample microbiological organisms from any facility system. Compressed air systems are unique; ISO and USP have select protocols specifically designed for the analysis of microbial contaminants, which utilize negative controls, blinds, and aseptic technique. When properly done, the results paint a clear image of the sampling day. If ISO 8573-7 is followed verbatim, there exists a sterility blank (a negative control that accompanies the test samples but is never opened), two blinds that test the aseptic technique of the technician, before and after sampling, and the actual point-of-use sample. Should any of the ISO 8573-7 blinds contain contamination, there is little confidence as to which system contaminated the test plates, should the test plate have contamination as well (the ambient environment or the compressed air). More specifically, if a before blind is contaminated with 2 colony forming units (CFU) and the point-of-use test plate is contaminated with 1 CFU, while the after blind is clear of observable contamination, there would now exist a level of scrutiny that must be placed on that point-of-use’s data. There can be no assurances that the contamination on the test plate is not the same as the contamination on the before blind. Likewise, if the blinds are contaminated but the compressed air sample is not, the aseptic training of the sampling technician should be reassessed and evaluated. The ideal situation is clean blinds and an under-limit test plate report.
In summary, there is no substitution for proper training of personnel for aseptic technique. The microbial results can indicate whether protocol was adequately followed or if retraining needs to occur. In either case, documentation and biannual training assessments can aid in maintaining technique and accurate compressed air microbial reports. The cost of contaminated blinds and compressed air microbial samples can result in retesting fees and labor costs, false positives that result in potential point-of-use shut downs and decrease in product integrity if a recall is issued.
About the Author
Maria Sandoval has over 15 years of experience in Microbiology and Molecular Biology. Her field work includes analyzing extremophiles isolated from the depths of Lake Baikal in Russia to the 50km exclusion zone of Chernobyl. Additionally, she’s worked alongside the CDC with DSHS analyzing and diagnosing patient microflora. Her tenure with the Lawrence Berkeley National Laboratory, Department of State Health Services and the University of Texas MD Anderson Cancer Center has made her a leading expert in microbial testing. As Trace Analytics’ Microbiologist and Lab Director, she is responsible for microbial testing and procedural development.
Trace Analytics is an A2LA accredited laboratory specializing in compressed air and gas testing for food and beverage manufacturing facilities. Using ISO 8573 sampling and analytical methods, their laboratory tests for particles (0.5-5 microns), water, oil aerosol, oil vapor, and microbial contaminants found in compressed air. For over 29 years, they’ve upheld the highest industry standards of health and safety, delivering uncompromising quality worldwide in accordance with ISO, SQF, BRC, and FDA requirements. Visit www.AirCheckLab.com
To read similar articles about Compressed Air Purification please visit www.airbestpractices.com/technology/air-treatment.
References
- Tamburini, E., Donega, V., Marchetti, M.G., et.al. (2017, July 17). Study on Microbial Deposition and Contamination onto Six Surfaces Commonly Used in Chemical and Microbiological Laboratories. Int. J. Environ Res Public Health. 12(7):8295-8311.
- Centers of Disease Control and Prevention. (n.d.) Show Me the Science- Why Wash Your Hands? Retrieved April 15, 2019 from, https://www.cdc.gov/handwashing/why-handwashing.html?s_cid=ostltsdyk_cs_679.
- Human Oral Microbiome Database. (n.d.) Human Oral Microbiome. Retrieved April 15, 2019 from, http://www.homd.org/.
- Bowler, J. (2017, June 20). Bacteria in your Coughs and Sneezes Can Stay Alive in the Air for up to 45 Minutes. Retrieved April 18, 2019 from, https://www.sciencealert.com/bacteria-in-your-coughs-and-sneezes-can-stay-alive-in-the-air-for-up-to-45-minutes.
- Brown, M.C., Conway, J., Sorensen, T.D. (2006, December 15). Development and Implementation of a Scoring Rubric for Aseptic Technique. Am J Pharm Educ. 70